Ready-to-go viral vector technology platform
exoREADY is a viral vector technology platform providing an off-the-shelf solution for transient expression for both adeno-associated virus (AAV) and lentivirus (LV) compatible with adherent and suspension culture based on a HEK-293 cell line. The platform also provides pre-optimized downstream purification processes, a development analytical toolkit and dedicated in-process and release quality control methods that can address all the steps required for the manufacture of complex advanced therapies in a fast, efficient, and lower cost manner.

Ready-to-go viral vector technology platform
exoREADY is a viral vector technology platform providing an off-the-shelf solution for transient expression for both adeno-associated virus (AAV) and lentivirus (LV) compatible with adherent and suspension culture based on a HEK-293 cell line. The platform also provides pre-optimized downstream purification processes, a development analytical toolkit and dedicated in-process and release quality control methods that can address all the steps required for the manufacture of complex advanced therapies in a fast, efficient, and lower cost manner.

Capitalize on exoREADY technology platform
Viral vector
dedicated platform
- EProprietary technology platform
- EDedicated to industrial manufacturing of viral vectors for Cell and Gene Therapy
- EFully customizable
- EHEK293 cell bank
scale-X™
bioreactor leader
- ENew industry gold standard
- EOne of the most experienced CDMOs
- ECompleted runs at Exothera: 60+
Ready to use
technology
- EState-of-the-art USP equipment
- EState-of-the-art DSP equipment
- EPre-developed analytical toolkit
- ECMC and regulatory support
Capitalize on exoREADY technology platform
Viral vector dedicated platform
- EProprietary technology platform
- EDedicated to industrial manufacturing of viral vectors for Cell and Gene Therapy
- EFully customizable
- EHEK293 cell bank
scale-X™ bioreactor leader
- ENew industry gold standard
- EOne of the most experienced CDMOs
- ECompleted runs at Exothera: 60+
Ready to use technology
- EState-of-the-art USP equipment
- EState-of-the-art DSP equipment
- EPre-developed analytical toolkit
- ECMC and regulatory support
Get faster to the clinical phase without quality compromise
We work day by day to reduce the development timeline without compromising on quality and costs.
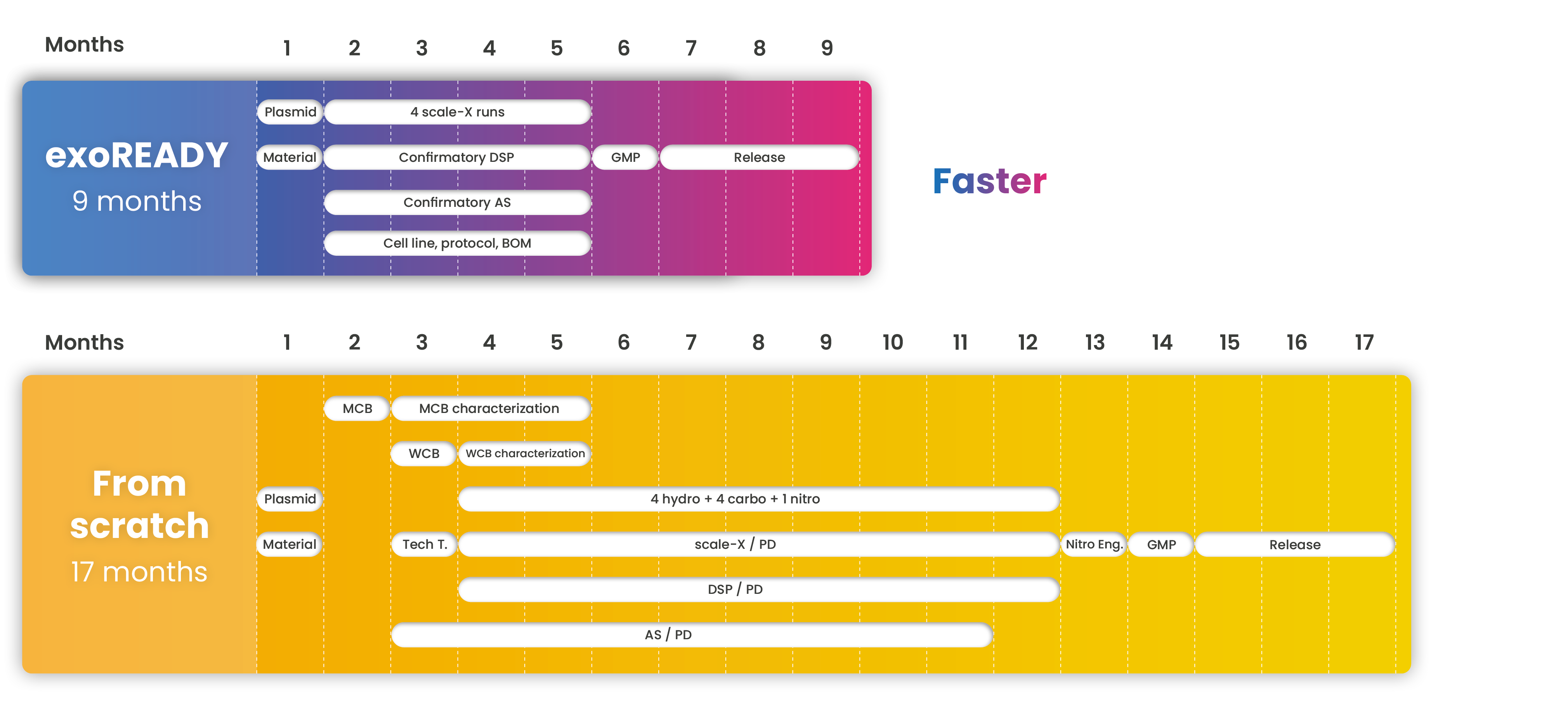
Get faster to the clinical phase without quality compromise
We work day by day to reduce the development timeline without compromising on quality and costs.



